New studies show that preserving lymph nodes may be critical for effective anti-tumour immune responses (Figure 1).
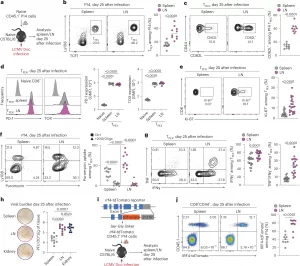
Figure 1: LNs preserve the stemness, proliferative fitness and polyfunctionality of T cells. a–g, Naive congenically marked (CD45.1+) P14 CD8+ T cells were adoptively transferred into naive wild-type (WT) (CD45.2+) mice, which were subsequently chronically infected with LCMV Doc. On day 25 after infection, CD8+ T cells in the spleen and pooled inguinal, brachial, axillary, cervical and mandibular LNs were analyzed using flow cytometry. a, Schematic of the experimental setup. b,c, Flow cytometry plots and quantification showing the frequencies of TPEX cells among P14 cells (n = 10) (b) and CD62L+ cells among TPEX cells (n = 9) (c). d, Representative histograms and quantification showing the expression of PD-1 and TOX in P14 TPEX cells in the spleen and LNs (n = 10). e, Flow cytometry plots and quantification showing Ki-67 expression in P14 TPEX cells in the spleen and LNs (n = 21). f, Flow cytometry plots and quantification showing puromycin incorporation in P14 cells in the spleen and LNs (n = 13). g, Flow cytometry plots and quantification showing IFNγ and TNF production in P14 TPEX cells from the spleen and LNs after stimulation with gp33 peptide (n = 19). h, Representative plaque assays and quantification showing viral titers in the spleen, LNs and kidney on day 25 after infection (n = 10). i,j, Naive congenically marked (CD45.1+) P14 CD8+ T cells expressing an IRF4-tdTomato fusion protein were adoptively transferred into naive WT (CD45.2+) mice, which were subsequently infected with LCMV Doc. On day 25 after infection, CD8+ T cells in the spleen and pooled inguinal, brachial, axillary, cervical and mandibular LNs were analyzed using flow cytometry. i, Schematic of the Irf4-tdTomato locus and experimental setup. j, Representative flow cytometry plots and quantification showing the expression of IRF4 in P14 cells in the spleen and LNs (n = 10). Flow cytometry plots are representative. b–h,j, The dots represent individual mice; the bars represent the median. Quantification and statistics were done using an unpaired two-tailed t-test (b,c,e,g,j) and a one-way analysis of variance (ANOVA) (d,f,h) and are based on all data points across at least two independent experiments. GMFI, geometric mean fold increase.
- Persist long-term during chronic disease
- Self-renew
- Generate waves of short-lived “killer” T cells that directly attack tumours or infected cells
- Stem-like T cells originating in lymph nodes are the primary drivers of successful immunotherapy
- T cells residing within tumours themselves are far less capable of sustaining long-term anti-tumour responses
- Removing lymph nodes can reduce the immune system’s capacity to respond, even when powerful immunotherapies are used
- Designing therapies that protect or enhance lymph node function
- Developing immunotherapies that better exploit lymph-node-derived immune cells
- Improving patient stratification based on immune architecture, not just tumour features
- Surgical strategies may need to balance cancer control with immune preservation
- Immunotherapies may be more effective when lymph node integrity is maintained
- Future treatments could deliberately target lymph nodes to amplify anti-tumour immunity