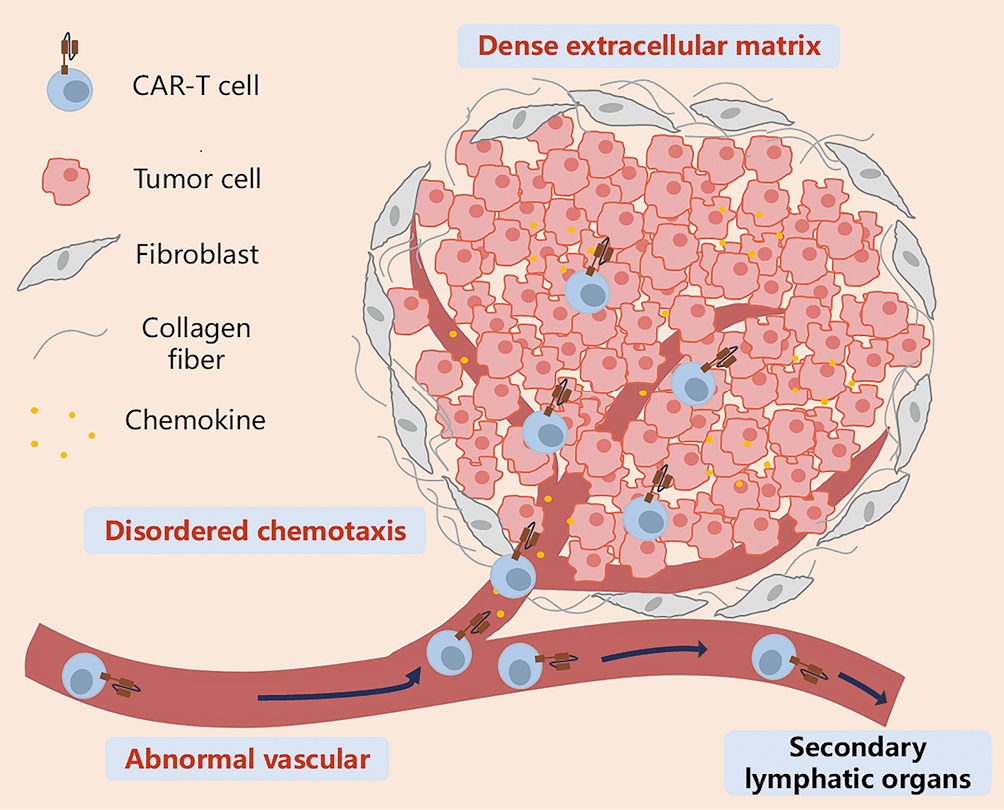
New strategies aim to overcome the biggest barrier to CAR-T success beyond blood cancers (Figure 1).
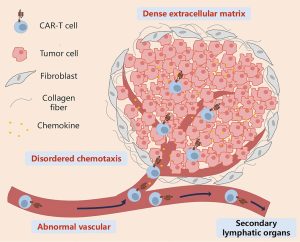
Figure 1: Key features that prevent CAR-T cells from infiltrating solid tumors through the circulation. CAR-T cells migrate to tumor tissues through the bloodstream under the guidance of chemokines, cross the abnormal tumor vasculature, penetrate the vicinity of tumor cells by overcoming barriers posed by the extracellular matrix and stromal cells, and ultimately generate stable intercellular contacts with tumor cells. During this process, disordered chemotaxis, abnormal tumor neovascularization, mesenchymal stromal cells dominated by cancer-associated fibroblasts, and a dense extracellular matrix are the main barriers that prevent CAR-T cells from effectively contacting tumor cells. CAR-T: Chimeric antigen receptor T.
- Abnormal blood vessels that limit immune cell trafficking
- A dense extracellular matrix (ECM) that acts like a physical wall
- Disrupted chemokine signals that fail to guide T cells to tumours
- Immunosuppressive stromal cells, including cancer-associated fibroblasts
- Secrete chemokines such as CCL19 or CXCL10, or
- Express matching chemokine receptors like CXCR6
- Targeting fibroblast activation protein (FAP) on cancer-associated fibroblasts
- Using enzymes such as hyaluronidase to degrade ECM components
- Employing synNotch CAR-T cells that release matrix-degrading enzymes only within the tumour
- Chemotherapy (e.g., nab-paclitaxel) to loosen stromal structure
- Radiotherapy to induce inflammatory signals
- Oncolytic viruses to reshape the tumour microenvironment
- Translating complex preclinical models into human success
- Optimizing CAR-T cell phenotypes for hostile solid-tumour environments
- Scaling biomaterial-based delivery strategies for clinical use