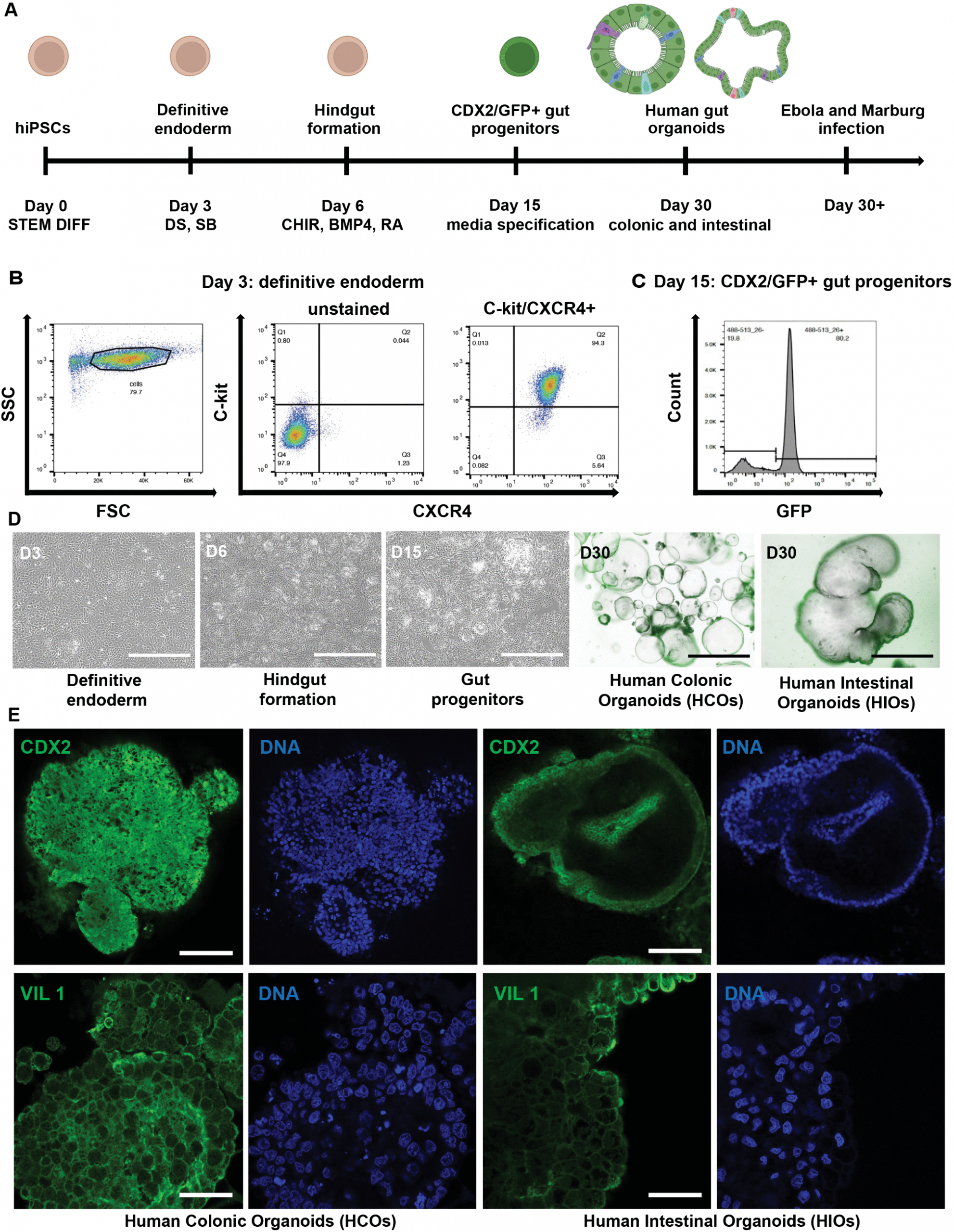
New research reveals that deadly filoviruses directly infect intestinal cells, disrupting fluid balance and driving life-threatening diarrhoea (Figure 1).
Figure 1: Directed differentiation of human iPSCs into region-specific intestinal (HIOs) and colonic (HCOs) organoids. (A) Schematic representation of the stepwise differentiation protocol used to generate gut organoids from human induced pluripotent stem cells (iPSCs). Illustrations created in BioRender. Muhlberger, E. (2025) https://BioRender.com/14baw98. (B) BU1 CDX2-GFP iPSCs were differentiated to definitive endoderm by day 3. (C) A CDX2-eGFP knock-in reporter line was used to monitor the emergence of hindgut progenitors during differentiation. (D) Gut progenitor cells were further specified into regional identities using defined media: colonic organoids (HCOs) were cultured in CKDCI media, and intestinal organoids (HIOs) were cultured in media containing CHIR, KGF, EGF, R-spondin, and Noggin. Brightfield and fluorescence imaging was performed at day 30 to visualize organoid morphology. Images were captured using a Keyence BZ-X710 fluorescence microscope. (E) Immunofluorescence staining was used to detect CDX2 and villin (VIL) (green) and Hoechst for nuclear staining (blue) in HIOs and HCOs. Confocal imaging was performed using a Zeiss LSM 710-Live Duo Confocal microscope with two-photon capability. Scale bars = 100 μm. Data shown are representative of n = 3 independent differentiations.
- Colon-like organoids showed more severe dysfunction than small-intestine-like organoids
- Key pathways controlling ion transport and fluid secretion were disrupted
- Structural damage occurred at the apical surface and cell–cell junctions, which normally regulate what passes through the intestinal wall
- Targeted supportive therapies to preserve gut barrier function
- Improved clinical management of fluid loss
- Antiviral strategies aimed at protecting epithelial tissues, not just immune cells