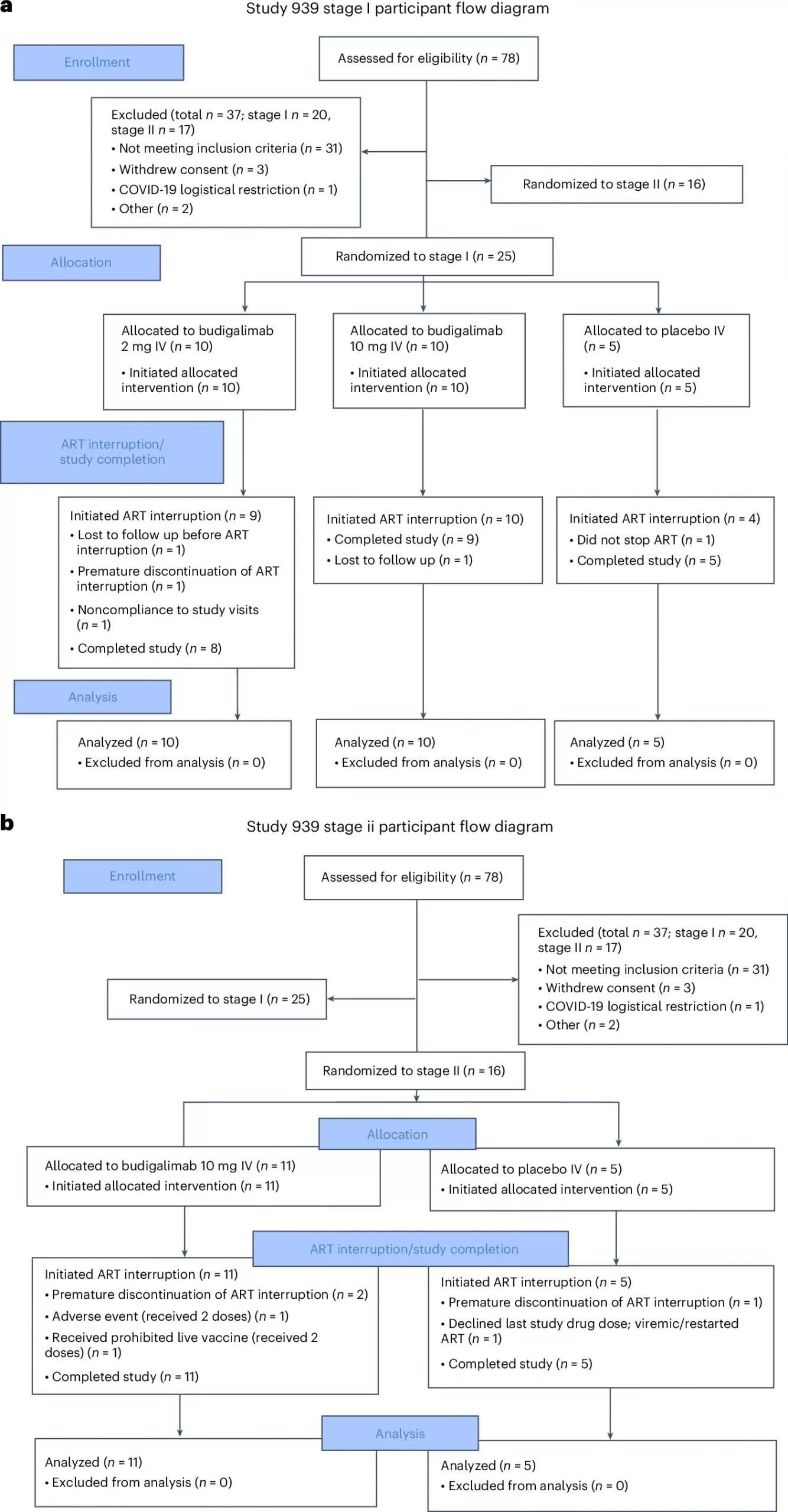
A treatment originally designed for cancer may hold promise for people living with HIV. A new Phase 1 clinical trial, reports that budigalimab, an immune checkpoint inhibitor targeting programmed cell death protein 1 (PD-1), was safe, well tolerated, and showed early signs of helping the immune system control HIV without daily medication (Figure 1).
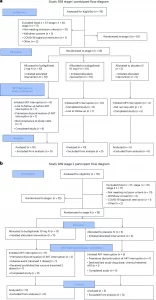
Figure 1: Full CONSORT participant flow diagram for Study M19-939 a) stage I and b) stage II. ART, antiretroviral therapy; IV, intravenous.
- Six of eleven participants receiving budigalimab in the second stage of the study experienced a delayed viral rebound, while
- Two participants maintained viral suppression for over six months without ART. No participants in the placebo group experienced this benefit.