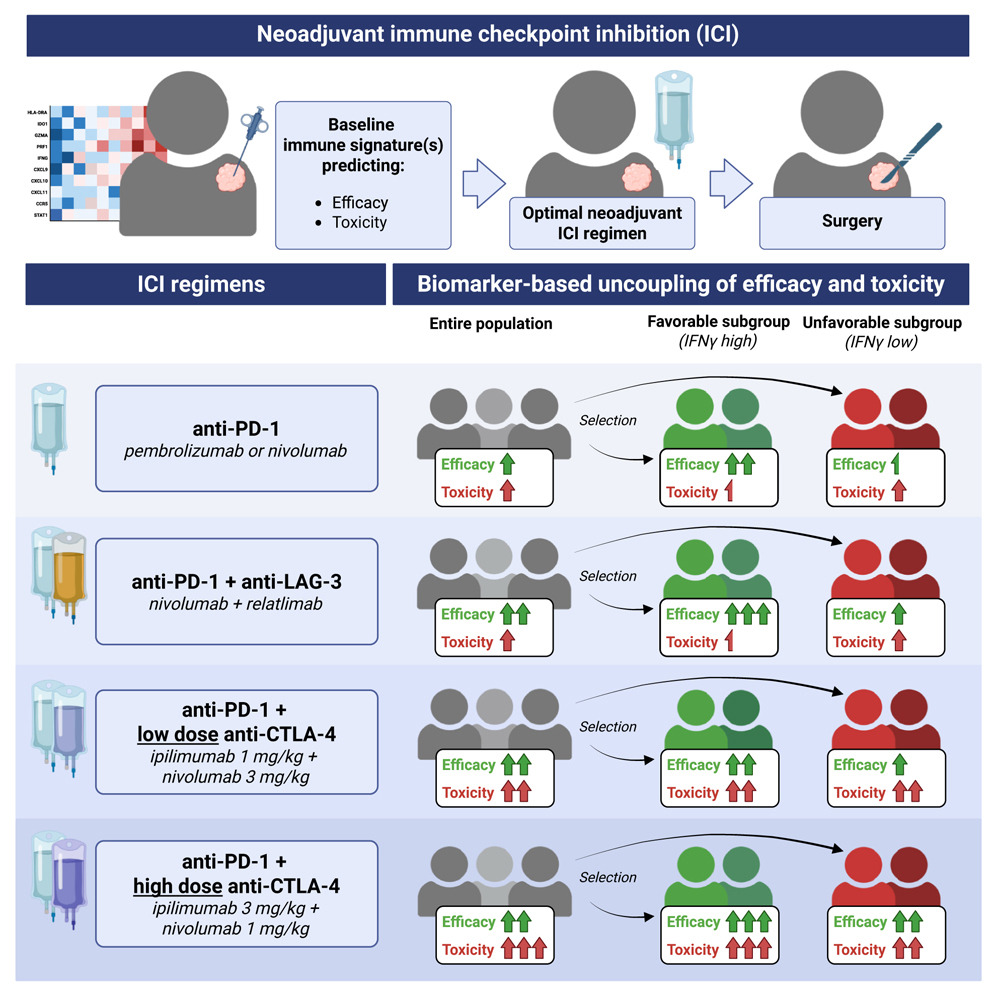
New analysis suggests immune signatures can guide safer, more effective checkpoint inhibitor combinations in melanoma (Figure 1).
Immune checkpoint inhibitors (ICIs) have transformed melanoma treatment, but not all patients benefit and many experience serious immune-related adverse events (irAEs). A long-standing challenge in the field has been balancing efficacy against toxicity, particularly when combining agents such as anti–PD-1 and anti–CTLA-4 therapies. A new report suggests this balance may be achievable through immune-guided personalization. By analysing how pathological response and toxicity relate to baseline immune activity, the researchers show that the same treatment can behave very differently depending on a patient’s immune state. The team conducted a meta-analysis of neoadjuvant melanoma trials, comparing four immune checkpoint regimens:- Anti–PD-1 alone
- Anti–PD-1 + low-dose anti–CTLA-4
- Anti–PD-1 + high-dose anti–CTLA-4
- Anti–PD-1 + anti–LAG-3
- In IFN-γ–high tumours, adding (especially high-dose) anti–CTLA-4 substantially increased toxicity but did not improve efficacy over anti–PD-1 alone.
- In contrast, in IFN-γ–low tumours, adding high-dose anti–CTLA-4 significantly improved response rates, while toxicity remained comparatively low.
- Does this patient need stronger immune activation to respond?
- Is this patient already primed and therefore more vulnerable to immune toxicity?
- Improve outcomes in patients with immune-cold tumours
- Reduce unnecessary toxicity in immune-hot patients
- Enable more rational sequencing of PD-1, CTLA-4, and emerging checkpoints like LAG-3