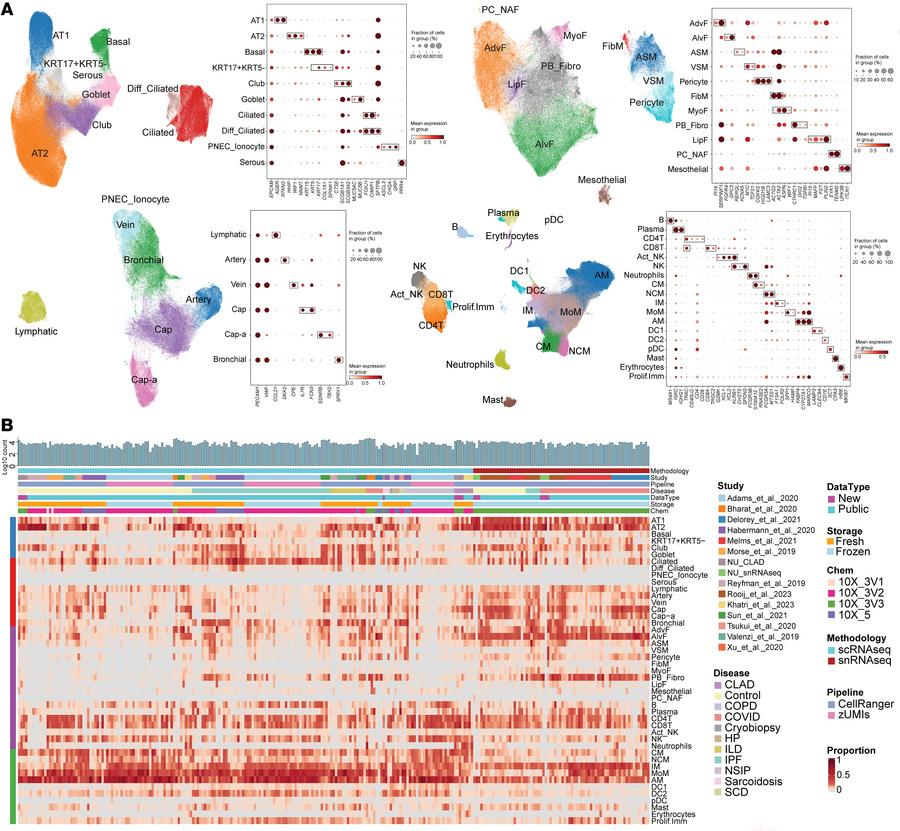
Lung transplantation offers a vital lifeline for patients with end-stage lung disease, yet more than 50% of lung-transplant recipients experience organ rejection within five years. Despite decades of research, the biological mechanisms driving this chronic rejection have remained elusive. Recently, scientists have provided the most detailed picture yet of how lung damage and transplant rejection unfold at the cellular level (Figure 1). Their study reveals that abnormal cross-talk between donor and recipient cells drives progressive scarring, leading to chronic rejection and organ failure.
Figure 1: Integrated object of single-cell transcriptome from multiple datasets. (A) Uniform manifold approximation and projection (UMAP) plot displaying cell types, accompanied by a dot plot showing the expression levels of key marker genes. The expression level was z-score–transformed. AMs, alveolar macrophages; IM, interstitial macrophages; MoM, monocyte-derived macrophages. (B) Heatmap of cell type proportions across samples, where each row represents a cell type, and each column represents a sample. Diff_Ciliated, differentiating ciliated; PNEC_Ionocyte, pulmonary neuroendocrine cells and ionocyte; ASM, airway smooth muscle; VSM, vascular smooth muscle; FibM, fibromyocyte; LipF, lipofibroblast; PC_NAF, perichondrial or nerve-associated fibroblast; Cap, general capillary endothelial cells; Cap-a, capillary aerocyte; NCM, nonclassical monocytes; Act_NK, activated NK; pDC, plasmacytoid dendritic cells; Prolif.Imm, proliferative immune cells.
- “Exhausted” T cells that remain chronically activated but lose their ability to regulate inflammation.
- “Super-activated” macrophages that persistently promote tissue damage and fibrosis.